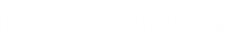Medicines and Healthcare products Regulatory Agency
815 articles
Showing 631-660

Dovonex Psoriasis Ointment to be available to buy from pharmacies
Thursday, 17 August 2017
Reclassification of Dovonex Psoriasis Ointment approved by MHRA...

MHRA successfully reclassifies Dovonex Psoriasis Ointment
Thursday, 17 August 2017
Medicines and Healthcare products Regulatory Agency (MHRA) have today announced the approval of Dovonex Psoriasis 50microgram/g Ointment, which will b...
Making safer polio vaccines with plants
Tuesday, 15 August 2017
A new study, involving scientists from National Institute for Biological Standards and Control (NIBSC), has reported the production of stable poliovir...

Speech given by Lord O’Shaughnessy on Brexit and medicines regulation
Friday, 11 August 2017
Speech given by Lord O’Shaughnessy on Brexit and medicines regulation, at the 2017 MHRA/BIA Conference on 14 July in London....

Professor Sir Michael Rawlins re appointed Chair
Tuesday, 08 August 2017
Professor Sir Michael will return as Chair for a second term....

MHRA response to the final report of the Mesh Oversight Group
Wednesday, 26 July 2017
NHS England set up the Mesh Working Group to address the concerns of a number of patients and clinicians....

Successful reclassification of Nasonex Allergy Control Nasal Spray
Monday, 10 July 2017
MHRA reclassifies Nasonex Allergy Control...

Nasal decongestant to be available to buy from pharmacies
Wednesday, 05 July 2017
MHRA reclassifies Otrivine Extra Dual Relief Nasal Spray Solution...

Maloff Protect successfully reclassified
Monday, 03 July 2017
After a public consultation, Maloff Protect will now be available over the counter in pharmacies...

Maloff Protect antimalarial tablets to be available to buy from pharmacies
Monday, 03 July 2017
Maloff Protect anti-malarial tablets will shortly be available to buy from pharmacies, after previously only being available to purchase with a prescr...

All types of metal on metal hip implants need regular review
Thursday, 29 June 2017
MHRA updates guidance for healthcare professionals....

MHRA chair recognised in 2017 Queen's Birthday Honours
Tuesday, 20 June 2017
Professor Sir Michael Rawlins, MHRA chair, honoured in the 2017 Queen's Birthday Honours List....
New SCOPE ADR e learning module receives European wide CME/CPD accreditation
Friday, 16 June 2017
A new e-learning module on the importance of reporting suspected side effects to medicines has received European accreditation....

Faulty defibrillator: users urged to check battery connection
Thursday, 18 May 2017
People and organisations should check public access defibrillators for a technical fault, the Medicines and Healthcare products Regulatory Agency advi...

Losing GSI – MHRA emails are changing
Monday, 10 April 2017
From 10 April, we are removing .gsi from our email addresses. ...

A new way to pay
Friday, 31 March 2017
Paying for medicines licences, clinical trials and clinical investigations is changing from 1 April the Medicines and Healthcare products Regulatory A...

Couple sentenced for selling dangerous slimming pills online
Friday, 31 March 2017
A husband and wife from Bootle have today been sentenced at Liverpool Crown Court for the illegal online sale of unlicensed slimming pills....
Check you’re selling medicines online lawfully
Friday, 17 March 2017
Online retailers must follow UK law, as well as the law of the country to which they are selling medicines...

New essential Orange and Green Guides 2017 – out now
Tuesday, 28 February 2017
The Medicines and Healthcare products Regulatory Agency has launched its latest guide of UK pharmaceutical regulations, EU directives and guidance. ...
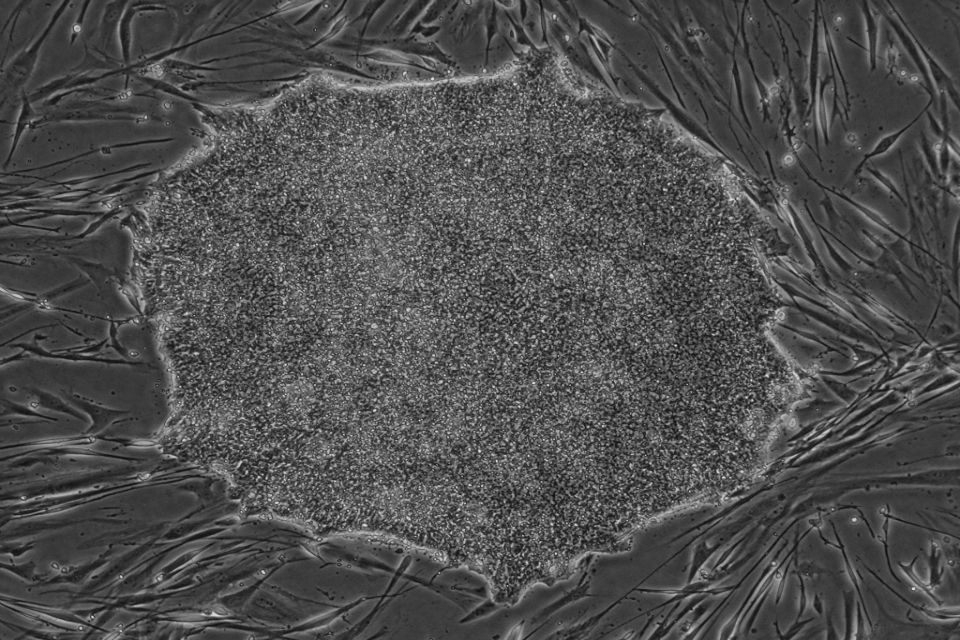
‘Regulator ready’ stem cell lines now available for clinical development
Monday, 27 February 2017
The UK Stem Cell Bank (UKSCB) at the National Institute for Biological Standards and Control (NIBSC) is releasing its first stem cell lines suitable f...

New MedRegs blog
Thursday, 09 February 2017
MHRA has launched an official blog providing expert insight into the latest regulatory thinking and all aspects of medicines regulation....

Oxford men admit to illegally supplying medicines
Wednesday, 08 February 2017
Investigation by the Medicines and Healthcare products Regulatory Agency (MHRA) results in guilty pleas for supplying unlicensed, and potentially dang...

MHRA warns athletes to avoid potentially dangerous DMAA
Monday, 30 January 2017
New Years warning issued urging athletes to avoid potentially dangerous DMAA, otherwise known as Methylhexanamine...

MHRA launch 'DMAA Week of Action'
Monday, 30 January 2017
MHRA has launched a ‘Week of Action’ to improve awareness of unlicensed medicines containing the potentially dangerous ingredient DMAA....

Standards for biological medicines understanding them and how they make a difference
Monday, 09 January 2017
The quality of biological medicines is assured by biological standards held by the British Pharmacopoeia (BP) and the National Institute for Biologica...

#FakeMeds in 2017: New Year, Same Danger
Tuesday, 03 January 2017
In a New Year with new beginnings, the dangers of fake and unlicensed medical products remain the same. We continue our campaign against fake and unli...

Agency highlights 2016
Friday, 30 December 2016
Looking back at what the Medicines and Healthcare products Regulatory Agency has achieved over the last year....

Regulator investigating the diversion of prescription only medicines
Friday, 16 December 2016
The Medicines and Healthcare products Regulatory Agency (MHRA) is conducting a large scale investigation into the diversion of prescription only medic...

Count down to Christmas with the FakeMeds advent calendar
Wednesday, 14 December 2016
MHRA are revealing a different fact about the issue of fake and unlicensed medical products every day in December leading up to Christmas....

Partnership to protect public health – MHRA and HTA sign agreement
Thursday, 08 December 2016
The Medicines and Healthcare products Regulatory Agency (MHRA) and the Human Tissue Authority (HTA) have today signed a partnership agreement....